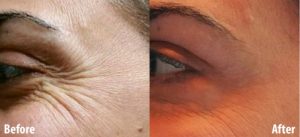
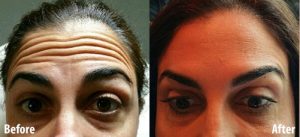
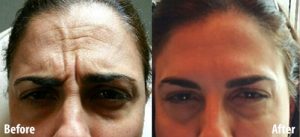
Botox
BOTOX® has been used safely and successfully in ophthalmology since 1989. It has been used for wrinkle therapy even before its 2002 FDA cosmetic approval. Only minute amounts of diluted toxin are injected for wrinkle reduction.

What are the overall advantages of treating wrinkles with BOTOX®?
- Temporarily reduces wrinkles without surgery.
- No sedation or anesthesia is needed.
- Affects underlying muscles (as opposed to the skin’s surface or
skin’s sub-layer the way peels do). - Reduces horizontal creases in the forehead. Reduces frown lines.
- Minimizes the look of crows’ feet and laugh lines.
- Effects are customizable.
- Some patients report they no longer get migraine headaches.
How does Botox work?
A few drops of BOTOX® are injected with a tiny needle into the muscle that creates a wrinkle. When BOTOX® is injected into a muscle, it blocks the nerve impulse from reaching that area, and as a result, the muscle weakens. As the muscle weakens, the skin overlying the muscle relaxes and the wrinkles in the skin gradually soften and often disappear. BOTOX® treatment does not effect normal facial expressions.
Botulinum Toxin Type A (BOTOX® Cosmetic) is a protein complex produced by the bacterium Clostridium botulinum, which contains the same toxin that causes food poisoning. When used in a medical setting as an injectable form of sterile, purified botulinum toxin, small doses block the release of a chemical called acetylcholine by nerve cells that signal muscle contraction. By selectively interfering with the underlying muscles’ ability to contract, existing frown lines are smoothed out and, in most cases, are nearly invisible in a week.
Basically, the botulinum toxins block the signals that would normally tell your muscles to contract. If a specific muscle can’t move, it can’t cause a wrinkle.
Treatment Procedure
- Your Practitioner will administer several tiny injections of BOTOX® Cosmetic directly into the muscles of the areas you wish treated.
- The location of the injections will be determined by examining your ability to move certain muscles in the selected area. The location, size, and use of the muscles that create a furrowed brow vary markedly among individuals.
- No anesthesia is required, although you may choose to have the area numbed with a cold pack or anesthetic cream prior to injecting. Discomfort is usually minimal and brief. Most patients compare the sensation to a pin prick and treatment takes only a few minutes.
- You may resume normal activity immediately. You may see a marked improvement in the moderate to severe frown lines between your brows within days. Improvement may continue for as long as several months, and could last up to 5 months*. Each patient has a different response. After that, the muscle action returns. When frown lines reappear, a repeat treatment is necessary to regain the results. * Results vary from patient to patient.
Dysport®
 DynaMIC muscle contraction is responsible for a number of undesirable lines and wrinkles on the face. For over a decade Botox has been the only product used to reduce dynaMICally caused wrinkling and lines. Now with Dysport both patients and doctors have a choice. (We love competition because it helps us get better pricing for our patients!)
DynaMIC muscle contraction is responsible for a number of undesirable lines and wrinkles on the face. For over a decade Botox has been the only product used to reduce dynaMICally caused wrinkling and lines. Now with Dysport both patients and doctors have a choice. (We love competition because it helps us get better pricing for our patients!)
How Dysport is different
Dysport is very similar to Botox with the primary neuroprotein, botulinum type A, being the same. The differences lie in the dosing unit calculation due to dilution type, the time it takes to see effectiveness, dispersion of the product and possibly longevity.
Dysport for many patients seems to take effect a little quicker than Botox, and for some reports lasts a little longer.
Who makes Dysport?
Dysport is distributed by Galderma (previously Medicis), the same company which now supplies Restylane and Perlane / Restylane Lyft. It was developed by Ipsen, a global pharmaceutical developer headquartered in France and has been used medically since 1991.
Xeomin®
Frown lines form when facial expressions are made as the muscle under the skin contracts. Over time, as your skin ages, these repeated expressions cause lasting frown lines. Neurotoxins, such as XEOMIN®, are prescription medications that block the release of chemicals that cause these muscle contractions so frown lines are softened.
XEOMIN® is made through a unique precision manufacturing process that isolates the therapeutic component of the molecule and removes the accessory proteins that don’t play an active role in treatment. XEOMIN® is a highly purified neurotoxin.
Studies have not been performed to determine whether the presence or absence of accessory proteins has a long-term effect on safety or efficacy.